Immunotherapy encompasses several different treatment approaches, each of which has a distinct mechanism of action, and all of which are designed to boost or restore immune function in some manner.
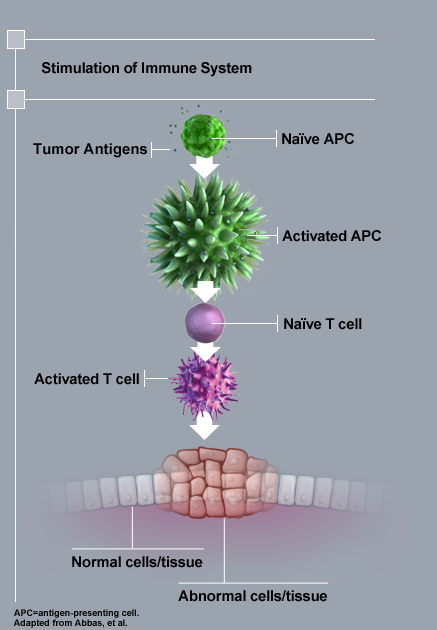
Therapeutic cancer vaccines are designed to stimulate the patient’s own immune system against tumor antigens. By triggering the immune system, therapeutic vaccines can initiate a durable anti-tumor response that can attack tumor cells and lead to improved survival. It is important to recognize, however, that therapeutic cancer vaccines differ from traditional preventative vaccines, such as those for various infectious diseases. Notably, the primary goal of a therapeutic cancer vaccine is NOT to prevent disease, but to generate an active immune response against an existing cancer.Some therapeutic cancer vaccines have contained a recombinant protein comprised of a tumor antigen and an immune cell activator. Once injected or infused, APCs process the antigen and then express antigenic fragments on their surface for presentation to the patient’s T cells. These activated APCs are able to interact with naïve T cells in the initiation of a T-cell driven immune response.
Tumor-specific monoclonal antibodies can elicit a direct or indirect immune response that leads to cell death. There are a variety of monoclonal antibodies and these work via different mechanisms of action to cause cell death.
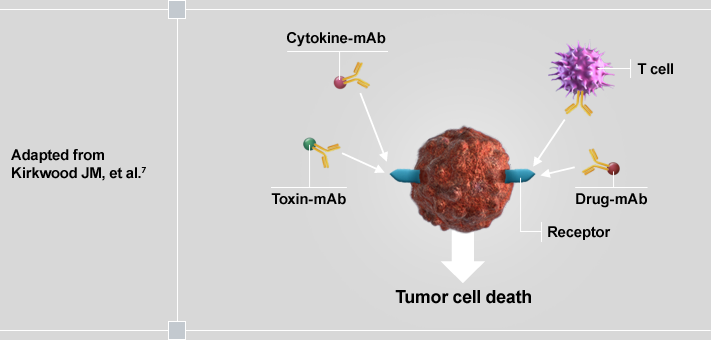
Some of these mechanisms include blocking signaling pathways needed for tumor cell growth, triggering an immune-mediated cytotoxic response (eg, antigen-dependent cellular cytotoxicity), or blocking angiogenesis.
Tumor-specific monoclonal antibodies have become part of the therapeutic repertoire in treating leukemia, breast, colorectal, and head and neck cancers after improving OS and progression-free survival in randomized, Phase 3 clinical trials. Response rates of 8%-10% have been observed when these are used as a single agent in advanced stage, heavily pretreated, and recurrent disease. These rates increase to 30% when combined with traditional chemotherapy and/or radiotherapy.
The immune system depends on multiple checkpoints or “immunological brakes” to avoid over activation of the immune system on healthy cells. Tumor cells often take advantage of these checkpoints to escape detection by the immune system. CTLA-4 and PD-1 are checkpoints that have been studied as targets for cancer therapy.
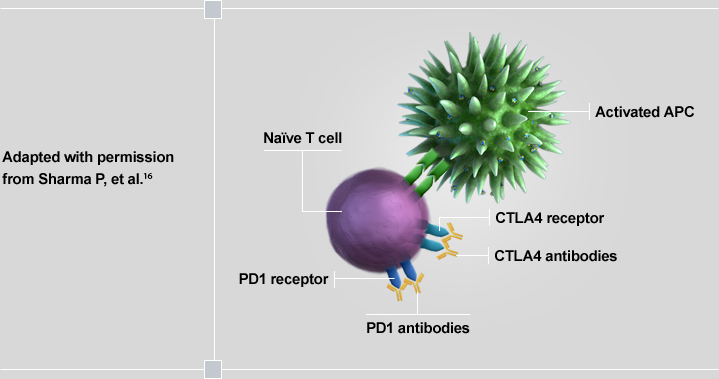
CTLA-4 has been shown to be aberrantly upregulated and present on the surface of T cells in certain cancers, dampening T-cell activation in response to tumor cells. PD-1 is another immunologic checkpoint that has been found to be upregulated in certain tumors; it inhibits T-cell function contributing to the tumor’s ability to evade the immune system. Inhibiting a checkpoint (ie, “releasing the brakes”) on the immune system may enhance the anti-tumor T-cell response. This class of therapy has shown efficacy in cancer and clinical trials are ongoing.
Cytokines, such as interleukin-2 (IL-2) and interferon-α (IFN-α), stimulate a broad-based immune response as opposed to generating a targeted response to a specific antigen. As an example, IL-2 has numerous effects on the immune system and acts as a general T-cell growth factor. It does this by binding to receptors on the surface of T cells.
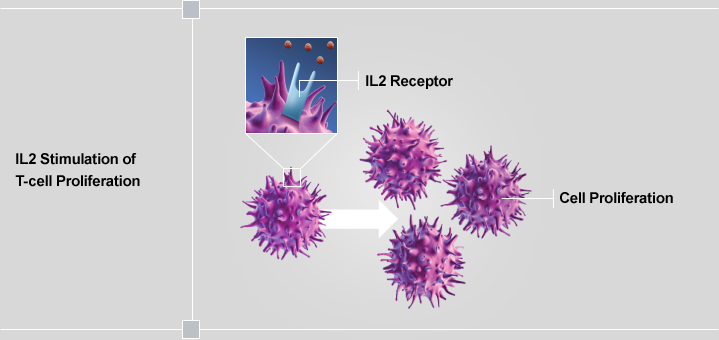
This binding stimulates the proliferation of T cells, continued cytokine production, and activation of multiple types of immune cells. It is interesting to note that high-dose IL-2 therapy has been shown to lead to a complete response in a subset of patients (4%-6%) in renal cell carcinoma and melanoma. This suggests that for this subset of patients, IL-2 therapy is able to successfully manipulate the endogenous anti-tumor immune response.